Chemistry, 05.05.2020 07:10, ghanim1963
An aqueous solution containing 9.82 g9.82 g of lead(II) nitrate is added to an aqueous solution containing 5.76 g5.76 g of potassium chloride. Enter the balanced chemical equation for this reaction. Be sure to include all physical states. balanced chemical equation: Pb(NO3)2(aq)+2KCl(aq)⟶PbCl2(s)+2KNO 3(aq)Pb(NO3)2(aq)+2KCl(aq)⟶PbCl2(s) +2KNO3(aq) What is the limiting reactant? potassium chloride lead(II) nitrate The percent yield for the reaction is 87.5%87.5% . How many grams of the precipitate are formed? precipitate formed: gg How many grams of the excess reactant remain?

Answers: 1
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 04:50, shonnybenskin8
Compare the equilibrium constants for the systems shown in the table. which favors products the most? which favors products the least? rank these systems in order from most to least in terms of favoring products rather than reactants. d > b > a > c c > a > b > d b > c > d > a a > d > c > b
Answers: 1

Chemistry, 22.06.2019 06:30, themajesty9898
The minerals found in bones are deposited by living cells called
Answers: 1

Chemistry, 22.06.2019 20:00, denaemarie02
Nitrogen dioxide decomposes according to the reaction 2 no2(g) ⇌ 2 no(g) + o2(g) where kp = 4.48 × 10−13 at a certain temperature. if 0.70 atm of no2 is added to a container and allowed to come to equilibrium, what are the equilibrium partial pressures of no(g) and o2(g)
Answers: 2

Do you know the correct answer?
An aqueous solution containing 9.82 g9.82 g of lead(II) nitrate is added to an aqueous solution cont...
Questions in other subjects:



History, 11.02.2021 01:00


Mathematics, 11.02.2021 01:00

Mathematics, 11.02.2021 01:00


Mathematics, 11.02.2021 01:00


Mathematics, 11.02.2021 01:00


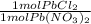 *
* 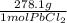 * 87.5/100 = 7.20 g PbCl₂
* 87.5/100 = 7.20 g PbCl₂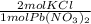 *
* 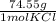 = 3.86 g KCl
= 3.86 g KCl