
Chemistry, 05.05.2020 11:05, mckinneypaige8243
A 14.3 g sample of HF is dissolved into 250 mL of solution. The concentration of the solution is *
A. 2.86 M
B. 0.14 M
C. 7.1 M
D. 3.6 M

Answers: 3
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 07:00, ceeejay0621
What is the main purpose of patent attorneys? defend the company against legal claims manage financial investments invent new products protect rights to new products and processes
Answers: 1



Chemistry, 23.06.2019 02:30, puppylover72
Calculate the ph at the equivalence point for the titration of a solution containing 150.0 mg of ethylamine (c2h5nh2) with 0.1000 m hcl solution. the volume of the solution at the equivalence point is 250.0 ml. kb forethylamine is 4.7 × 10−4 .
Answers: 2
Do you know the correct answer?
A 14.3 g sample of HF is dissolved into 250 mL of solution. The concentration of the solution is *
Questions in other subjects:

History, 18.08.2020 22:01

Chemistry, 18.08.2020 22:01

History, 18.08.2020 22:01






Social Studies, 18.08.2020 22:01

Mathematics, 18.08.2020 22:01


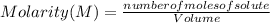
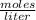 ).
).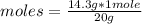
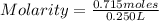
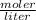 =2.86 M
=2.86 M



