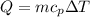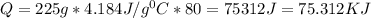Answers: 2
Other questions on the subject: Chemistry

Chemistry, 21.06.2019 19:30, aedmund1225
Agas has a volume of 0.7 l at 300 mmhg. what would be the new volume at 900 mmhg
Answers: 1

Chemistry, 22.06.2019 00:00, chameleonsarelife
Select the correct answer. which statement is true about a polarized object? o a. it gains electrons and becomes negatively charged. ob. it gains protons and becomes positively charged. oc. the number of positive and negative charges can be the same. od. it has to be a metal. o e. there is no change in the distribution of the charge in the object. reset next what
Answers: 3

Chemistry, 22.06.2019 04:30, anthony4034
Use the periodic table to determine the electron configuration of dysprosium (dy) and americium (am) in noble-gas notation.
Answers: 1

Chemistry, 22.06.2019 05:00, smartboy2296
Use the table to identify the phase and phase changes of the elements under the given conditions. write the name of the substance, phase, or phase change
Answers: 3
Do you know the correct answer?
A student must use 225 g of hot water in a lab procedure. Calculate the amount of heat in joules req...
Questions in other subjects:



Mathematics, 29.01.2020 12:55


Mathematics, 29.01.2020 12:55

Computers and Technology, 29.01.2020 12:55

English, 29.01.2020 12:55

Social Studies, 29.01.2020 12:55



 ) = 100°C - 20°C = 80°C, Water’s specific heat capacity (
) = 100°C - 20°C = 80°C, Water’s specific heat capacity (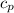 ) = 4.184 J/g°C
) = 4.184 J/g°C