
Chemistry, 05.05.2020 17:57, mooneyhope24
The standard molar enthalpy of formation of NH3(g) is -45.9 kJ/mol. What is the enthalpy change if 9.51 g N 2(g) and 1.96 g H2(g) react to produce NH
3(8)?
a.-29.8 kJ/mol-rxn
b.-43.7 kJ/mol-rxn
C.-10.3 kJ/mol-rxn
d.-20.7 kJ/mol-rxn
e. -65.6 kJ/mol-rxn

Answers: 2
Other questions on the subject: Chemistry


Chemistry, 22.06.2019 05:30, NorbxrtThaG
The table describes how some substances were formed substance 19 description formed by boiling pure water formed by combining three hydrogen atoms to every nitrogen atom formed by adding 5 g of sugar to 1 l of water formed by compressing carbon under high pressure based on the given descriptions, which substance is most likely a mixture?
Answers: 1


Chemistry, 22.06.2019 20:00, Isaiahtate053
The volume of a single vanadium atom is 9.29×10-24 cm3. what is the volume of a vanadium atom in microliters?
Answers: 3
Do you know the correct answer?
The standard molar enthalpy of formation of NH3(g) is -45.9 kJ/mol. What is the enthalpy change if 9...
Questions in other subjects:

History, 26.01.2021 21:00



Mathematics, 26.01.2021 21:00




Mathematics, 26.01.2021 21:00

Arts, 26.01.2021 21:00

Mathematics, 26.01.2021 21:00


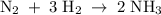
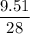
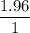
 2 moles of ammonia.
2 moles of ammonia.



