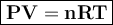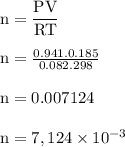Chemistry, 13.01.2020 01:31, natetheman7740
Chlorine can be prepared in the laboratory by the reaction of manganese dioxide with hydrochloric acid, hcl(aq), as described by the chemical equation.
how much mno2(s) should be added to excess hcl(aq) to obtain 185 ml of cl2(g) at 25 °c and 715 torr?
chlorine can be prepared in the laboratory by the reaction of manganese dioxide with hydrochloric acid, hcl(aq), as described by the chemical equation.
how much mno2(s) should be added to excess hcl(aq) to obtain 185 ml of cl2(g) at 25 °c and 715 torr?

Answers: 1
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 00:10, mpchop
According to the diagram; a) identify the anode of the cell and write the half-reaction that occurs there b) write the overall equation for the reaction that occurs as the cell operates c) calculate the value of the standard cell potential ,e cell. d)write the shorthand notation of the cell above e)indicate the flow of the electrons on the diagram
Answers: 3


Chemistry, 23.06.2019 03:30, uniqueray33
The molar mass of nickel(ni) is 58.7 g/mol. how many moles are in an 88 gram sample of nickel?
Answers: 1

Chemistry, 23.06.2019 10:10, nancysue1975
Calculate the h3o+ concentration in a solution of acetic acid if the concentration of molecular acetic acid present at equilibrium is 9.97x10-3 m and k for the dissociation is 1.86x10-5. ch3cooh(aq)+h2o(l)+> h3o+(aq)+ch3coo-(aq) show me how to get the answer.
Answers: 3
Do you know the correct answer?
Chlorine can be prepared in the laboratory by the reaction of manganese dioxide with hydrochloric ac...
Questions in other subjects:


Mathematics, 02.11.2020 19:30

Mathematics, 02.11.2020 19:30

Mathematics, 02.11.2020 19:30


Chemistry, 02.11.2020 19:30

English, 02.11.2020 19:30


Biology, 02.11.2020 19:30

Mathematics, 02.11.2020 19:30