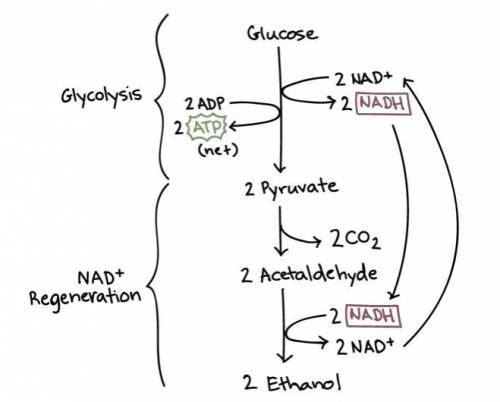
In yeast, ethanol is produced from glucose under anaerobic conditions. A cell‑free yeast extract is placed in a solution that contains 325 mmol glucose, 0.35 mmol ADP , 0.35 mmol P i , 0.70 mmol ATP , 0.20 mmol NAD + , and 0.20 mmol NADH . It is kept under anaerobic conditions. What is the maximum amount of ethanol (in millimoles) that could theoretically be produced under these conditions?

Answers: 2
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 03:30, tbeck225
If a solution is considered basic, then a) the hydroxide ion and hydronium ion concentrations are equal. b) the hydroxide ion concentration is less than the hydronium ion concentration. c) the hydronium ion concentration is greater than the hydroxide ion concentration. d) the hydroxide ion concentration is greater than the hydronium ion concentration.
Answers: 1


Chemistry, 22.06.2019 10:10, veronica022
Stage in which a typical star has completely stopped fusion
Answers: 1

Do you know the correct answer?
In yeast, ethanol is produced from glucose under anaerobic conditions. A cell‑free yeast extract is...
Questions in other subjects:



Arts, 24.01.2020 19:31

Biology, 24.01.2020 19:31

History, 24.01.2020 19:31





English, 24.01.2020 19:31