8. On the following picture, identify each of the following:
reactants
products
En...

Chemistry, 06.05.2020 02:12, Falconpride847
8. On the following picture, identify each of the following:
reactants
products
Energy
energy of reactants
energy of products
activation energy of forward reaction
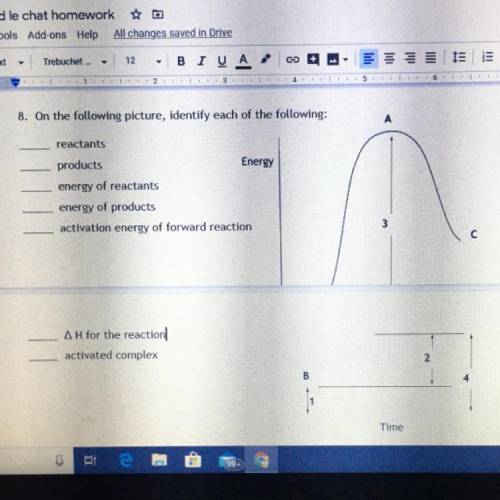

Answers: 3
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 15:20, Tringirl233
Identify arrows pointing to bonding electrons. done h-0-0-h ) intro
Answers: 3

Chemistry, 22.06.2019 18:00, LuvieAnn1886
How is energy related to the change of state represented by the model? atoms gain energy as a solid changes to a liquid. atoms gain energy as a solid changes to a gas. atoms lose energy as a solid changes to a liquid. atoms lose energy as a solid changes to a gas.
Answers: 3

Chemistry, 22.06.2019 22:20, icantspeakengles
Asuspension of yeast cells is being grown under anaerobic conditions such that glucose is degraded to ethanol and carbon dioxide. if one wishes to follow this process by monitoring the release of 14co2, at which positions in the glucose molecule would the 14c label need to be incorporated?
Answers: 2

Chemistry, 22.06.2019 22:40, hannahkharel2
Maleic acid is a carbon-hydrogen-oxygen compound used in dyeing and finishing fabrics and as a preservative of oils and fats.* in a combustion analysis, a 1.054-g sample of maleic acid yields 1.599 g of co_2 and 0.327 g of h2o.* when 0.609 g of maleic acid is dissolved in 25.00 g of glacial acetic acid, the freezing point drops by 0.82 c. maleic acid does not ionize in glacial acetic acid, which has a freezing-point depression constant of k_{\rm f}=3.90~^\circ {\rm c}/m.* in a titration experiment, a 0.4250- g sample of maleic acid is dissolved in water and requires 34.03 ml of 0.2152 m koh for its complete neutralization.* the ph of 0.215 g of maleic acid in 50.00 ml of aqueous solution is found to be 1.80.part adetermine the empirical formula for maleic acid. enter the atoms in order ofc, h, and o. part bdetermine the molecular formula for maleic acid. enter the atoms in order of c, h, and o. express your answer as a chemical formula. part cbased on the titration data, how many ionizable h atoms are in each molecule of maleic acid, c4h4o4? express your answer as an integer. i got the part a and b which is cho and c4h4o4 respectively, can anyone me for part c? the below is what i have so far from other qa'sfinding the empirical formula for maleic acid, needs this info"a combustion analysis a 1.054-g sample of maleic acid yields 1.599g of and 0.327g of ".the rest of your data is used to find molar mass, molecular formula, equivalent mass, # of h+ ions / molecule & katypically this should be: a 1.054-g sample of maleic acid yields 1.599g of co2 and 0.327g of h2osofind mass using molar masses1.599g of co2 @ (12.01 g carbon) / (44.01 g co2) = 0.436 grams of carbon0.327g of h2o @ (2.016 g hydrogen) / (18.01 g/mol h2o) = 0.037 grams of hydrogen1.054-g sample - 0.436 grams of carbon - 0.037 grams of hydrogen = 0.581 grams of oxygennow use molar masses to find moles0.436 grams of carbon @ 12.01 g/mol = 0.0363 mol c0.037 grams of hydrogen @ 1.008 g/mol = 0.0367 mol h0.581 grams of oxygen @ 16.00 g/mol = 0.0363 mol oyour empirical formula ischo and molecular is c4h4o4
Answers: 3
Do you know the correct answer?
Questions in other subjects:

Mathematics, 25.07.2019 09:40



Mathematics, 25.07.2019 09:40

Mathematics, 25.07.2019 09:40



Mathematics, 25.07.2019 09:40

Computers and Technology, 25.07.2019 09:40

Computers and Technology, 25.07.2019 09:40






