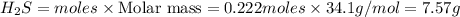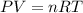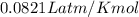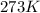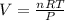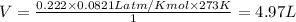Chemistry, 06.05.2020 06:12, harleyandpope90
If 25.0 grams of Sb2S3 reacts with an excess of hydrochloric acid, how many grams of H2S are formed? What volume does the H2S formed occupy under conditions of STP?

Answers: 1
Other questions on the subject: Chemistry

Chemistry, 21.06.2019 22:30, Aidanjsauer
Check the correct box to describe the periodic trends in electronegativity. electronegativity across a period: decreases. increases. electronegativity down a group: decreases. increases.
Answers: 2

Chemistry, 22.06.2019 05:40, yah2muchh
Calculate: select the worksheet tab. this tab you calculate the analyte concentration. fill in the first set of boxes ("moles h2so4" and "moles naoh") based on the coefficients in the balanced equation. (if there is no coefficient, the value is 1.) record the appropriate volumes in the "ml naoh" and "ml h2so4" boxes. record the concentration of the titrant in the m naoh box. click calculate. what is the concentration listed
Answers: 2

Chemistry, 22.06.2019 12:20, missayers172
Achemistry student weighs out 0.306 g of citric acid (h3c6h5o7), a triprotic acid, into a 250 ml volumetric flask and dilutes to the mark with distilled water. he plans to titrate the acid with 0.1000 m naoh solution. calculate the volume of naoh solution the student will need to add to reach the final equivalence point. be sure your answer has the correct number of significant digits.
Answers: 3
Do you know the correct answer?
If 25.0 grams of Sb2S3 reacts with an excess of hydrochloric acid, how many grams of H2S are formed?...
Questions in other subjects:


Mathematics, 08.10.2021 14:00


Arts, 08.10.2021 14:00

Mathematics, 08.10.2021 14:00

History, 08.10.2021 14:00


Mathematics, 08.10.2021 14:00

Mathematics, 08.10.2021 14:00

History, 08.10.2021 14:00


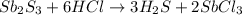
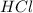 is the excess reagent,
is the excess reagent, 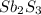 is the limiting reagent and it limits the formation of product.
is the limiting reagent and it limits the formation of product.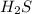
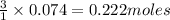 of
of