Chemistry, 13.01.2020 12:31, mayachojnicki
Including all coefficients, charges, and phases, what is the correct net ionic equation for the following set of reactants? assume that the contribution of protons from h2so4 is near 100% . ba(oh)2(aq)+h2so4(aq)
express your answer as a chemical equation.

Answers: 2
Other questions on the subject: Chemistry


Chemistry, 22.06.2019 13:30, yasiroarafat12
How many moles is 14.5 cm^3 of platinum? the density of platinum is 21.45 g/cm^3.
Answers: 1

Do you know the correct answer?
Including all coefficients, charges, and phases, what is the correct net ionic equation for the foll...
Questions in other subjects:





English, 01.04.2021 22:40



Mathematics, 01.04.2021 22:40


English, 01.04.2021 22:40



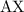 and
and 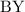 is as follows:
is as follows:
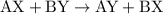
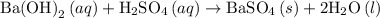
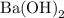 is
is 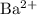 and
and 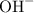 respectively. Similarly, the ionic species in
respectively. Similarly, the ionic species in 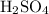 is
is 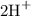 and
and 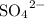 respectively. Hence the net ionic equation can be represented as follows:
respectively. Hence the net ionic equation can be represented as follows: