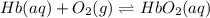As climbers ascend (go up) a mountain, the amount of oxygen in the atmosphere
decreases. In the...

Chemistry, 23.04.2020 23:18, lovethenae12
As climbers ascend (go up) a mountain, the amount of oxygen in the atmosphere
decreases. In the body, hemoglobin (Hb) in red blood cells binds with oxygen (02) to
form oxyhemoglobin (HbO2).
This equation can be used to model the equilibrium between hemoglobin , oxygen,
and oxyhemoglobin.
Hb + O2 <--> HbO2
According to Le Chatelier, how would the equilibrium shift as the climbers ascend to
higher altitudes?

Answers: 1
Other questions on the subject: Chemistry



Chemistry, 22.06.2019 05:40, Maryjasmine8001
Salicylic acid is a very important acid. it is used to synthesize the aspirin by treating with acetic anhydride. a 0.2015-g sample of salicylic acid was dissolved in a 100.00-ml volumetric flask, and the solution was diluted to the mark. a 10-ml aliquot of this solution was titrated with standard naoh (0.01130 + 0.2% n) to a phenolphthalein faint pink color end point at 19.81 ml. (a) (calculate the normality of the salicylic acid solution used in the titration. (b) assuming the salicylic acid is pure, what is the equivalent weight of the salicylic acid? practice problems for the final exam (continued) (c) (calculate the inherent error in the determination of the equivalent weight you calculated in part (b). use the following absolute errors in the equipment /glassware when calculating the inherent error. 5.00-ml pipet: + 0.02 ml 100-ml volumetric flask: + 0.08 ml analytical balance: + 0.2 mg 25-ml buret: + 0.03 ml
Answers: 2

Chemistry, 22.06.2019 09:30, strevino9178
In apex! a liquid heated beyond a certain temperature becomes
Answers: 1
Do you know the correct answer?
Questions in other subjects:



Mathematics, 04.10.2019 23:20


History, 04.10.2019 23:30



History, 04.10.2019 23:30


Mathematics, 04.10.2019 23:30