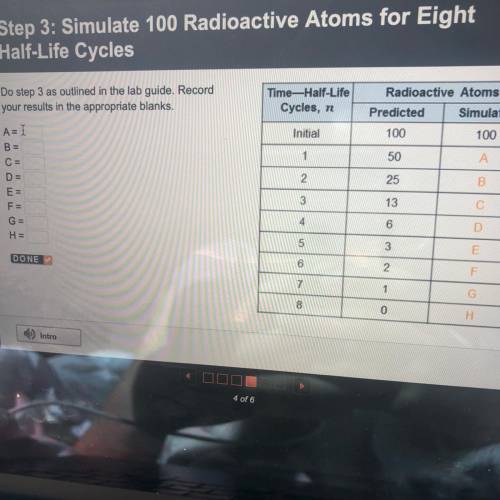
Do step 3 as outlined in the lab guide. Record
your results in the appropriate blanks.
T...

Chemistry, 23.04.2020 19:25, Delgadojacky0206
Do step 3 as outlined in the lab guide. Record
your results in the appropriate blanks.
Time-Half-Life
Cycles, n
Radioactive Atoms
Predicted Simulated
100
100
Initial
1
50
А
I
25
IOT MOOD
I
2
3
5
2
DONE

Answers: 3
Other questions on the subject: Chemistry


Chemistry, 22.06.2019 08:30, Apple557
The characteristic of two different types of reactions are shown below. reaction a: electrons are gained by the atoms of an element. reaction b: protons are lost by the atom of and element. which statement is true about the atoms of the elements that participate in the two reactions? a: their identity changes in both reaction a and b. b: their identity changes in reaction a but not b. c: their identity changes in reaction b but not a. d: their identity remains the same.
Answers: 1


Chemistry, 22.06.2019 12:10, yootmytoot
Achemistry student needs to standardize a fresh solution of sodium hydroxide. he carefully weighs out of oxalic acid , a diprotic acid that can be purchased inexpensively in high purity, and dissolves it in of distilled water. the student then titrates the oxalic acid solution with his sodium hydroxide solution. when the titration reaches the equivalence point, the student finds he has used of sodium hydroxide solution. calculate the molarity of the student's sodium hydroxide solution. be sure your answer has the correct number of significant digits.
Answers: 1
Do you know the correct answer?
Questions in other subjects:

Mathematics, 23.03.2020 19:57







Mathematics, 23.03.2020 19:57








