

Answers: 2
Other questions on the subject: Chemistry




Chemistry, 22.06.2019 15:00, emmalie52
Phosphorous acid, h3po3(aq) , is a diprotic oxyacid that is an important compound in industry and agriculture. p k a1 p k a2 1.30 6.70 calculate the ph for each of the points in the titration of 50.0 ml of 1.5 m h3po3(aq) 1.5 m h 3 po 3 ( aq ) with 1.5 m koh(aq). 1.5 m koh ( aq ) .
Answers: 1
Do you know the correct answer?
1) How many molecules are there in 985 mL of nitrogen at 0.0° C and 1.00 x 10-6 mm Hg?...
Questions in other subjects:

Geography, 23.01.2020 07:31






Chemistry, 23.01.2020 07:31




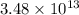
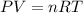
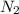 gas =
gas = 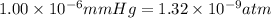 (1 atm = 760 mmHg)
(1 atm = 760 mmHg)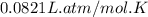
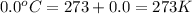
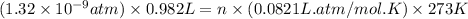
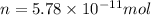
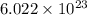 number of molecules.
number of molecules.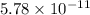 mole of
mole of 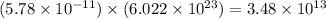 number of molecules
number of molecules




