
Chemistry, 23.04.2020 01:13, marbuigues9171
A saturated solution of barium fluoride, BaF2BaF2, was prepared by dissolving solid BaF2BaF2 in water. The concentration of Ba2+Ba2+ ion in the solution was found to be 7.52×10−3 MM . Calculate KspKspK_sp for BaF2BaF2.

Answers: 3
Other questions on the subject: Chemistry



Chemistry, 22.06.2019 22:30, itsmaddierae11
Which of the following molecules is polar? c3h7oh c2h5cooh
Answers: 1

Chemistry, 23.06.2019 00:00, bryn2433
Predict the relative bond lengths of the three carbon-oxygen bonds in the carbonate ion (co2−3). what would you expect the charge to be on each oxygen? match the words in the left column to the appropriate blanks in the sentences on the right. make certain each sentence is complete before submitting your answer.
Answers: 3
Do you know the correct answer?
A saturated solution of barium fluoride, BaF2BaF2, was prepared by dissolving solid BaF2BaF2 in wate...
Questions in other subjects:



Social Studies, 27.04.2021 23:20




Chemistry, 27.04.2021 23:20



Social Studies, 27.04.2021 23:20


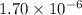
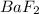 is given as:
is given as: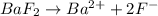
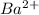 will be S moles/liter and solubility of
will be S moles/liter and solubility of  will be 2S moles/liter.
will be 2S moles/liter.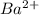 and 2 moles of
and 2 moles of ![K_{sp}=[Ba^{2+}][F^{-}]^2](/tpl/images/0620/0974/7de9f.png)
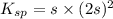
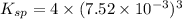
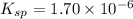
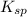 for
for 



