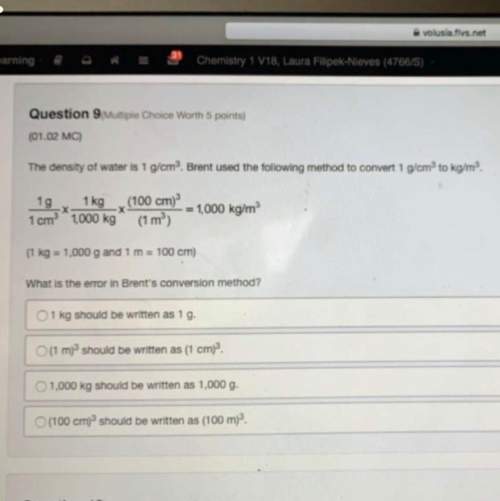
Chemistry, 22.04.2020 22:55, mckennacwilliams
For the reaction NO(g) + O3(g) ↔ NO2(g) + O2(g) at a temperature of 298 K, the standard Gibbs free energy change is ΔGº = -198 kJ/mol. Calculate the non-standard Gibbs free energy ΔG in kJ/mol when the partial pressures of these gases are those shown below.
Species Partial pressure in atm
NO(g) 1.0 x 10-8
O3(g) 1.0 x 10-9
NO2(g) 1.0 x 10-6
O2(g) 0.20
I am most interested in what form of R I should use, 0.008314 kj or 8.314 J
And what Q is??? Should I leave out pure elements when calculation Q?
Thanks!

Answers: 2
Other questions on the subject: Chemistry



Do you know the correct answer?
For the reaction NO(g) + O3(g) ↔ NO2(g) + O2(g) at a temperature of 298 K, the standard Gibbs free e...
Questions in other subjects:

Mathematics, 23.02.2021 09:50



Mathematics, 23.02.2021 09:50


Mathematics, 23.02.2021 09:50

Social Studies, 23.02.2021 09:50

Mathematics, 23.02.2021 09:50