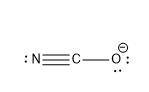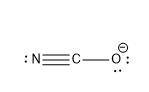The atom of oxygen
Explanation:
To understand better this, let's write the electron configuration for each element in the isocyanate ion, based on their atomic numbers:
Carbon = 6
Nitrogen: 7
Oxygen: 8
[C] = 1s² 2s² 2p²
[N] = 1s² 2s² 2p³
[O] = 1s² 2s² 2p⁴
Now that we have written this, let's figure out the number of electrons that each element can support. This can be seen in the last energy level of the configuration, in this case, the number 2. For the carbon, we can see that we have 4 electrons, nitrogen has 5 and oxygen has 6.
Now, how are they bonded? well, in this case, oxygen and nitrogen are bonded to the Carbon (The less electronegative element of them) and they are bonded by covalent bonds (This means that their united by electron sharing). In this case the carbon, with it's 4 electrons will unite on one side with the oxygen, and the other side the nitrogen. As Carbon has 4 electrons to share, 3 of these electrons are shared with the 3 of the nitrogen, and the remaining electron is bonded to the oxygen. (See picture attached).
Now, to know which element has the formal negative charge, we need to take account two things. the first thing is just need to apply the octect rule, that states that every element in the lewis structure, should have 8 electrons around it. To know this, we just count the electrons that were unpaired, and the electrons paired. The second thing is just count the individual electrons of each element, and this number should match the number of electrons that it should have. If for instance, we count the electrons of carbon and has 5 instead of 4, this means that it has 1 more electron, and this would have the formal negative charge.
See picture for this part. In the case of carbon, we can see it has 4 bonds (One triple bond and a single bond) and in total it has 8, so it has the octect. The nitrogen has 6 in the triple bond and 2 lone pair, so it has the octect. the oxygen has one bond, and 3 lone pairs, the octect.
Now let's see which of this elements has all it's electrons complete. In the case of carbon, we can see that it has 4 electrons shared, in the case of nitrogen, 3 of it's electrons are shared and 2 are not. but it has it's 5 electrons so it's complete. But in the case of oxygen, it has one electron bonded, but it also has 6 electrons unbonded, to give a total of 7. therefore it has one more electron, and that's why this would have the formal negative charge.
See picture for lewis structure