
Chemistry, 22.04.2020 22:08, tdahna0403
Carbon monoxide and chlorine combine in an equilibrium reaction to produce the highly toxic product, phosgene (COCl2 ) CO(g) Cl2 (g) COCl2 (g) If the equilibrium constant for this reaction is Kc = 248, predict, if possible, what will happen when the reactants and product are combined with the concentrations shown. [CO] = [Cl2] = 0.010 M; [COCl2] = 0.070 M

Answers: 3
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 00:10, scottbrandon653
Think about how you can use le chatelier’s principle to find possible solutions to the design problem. describe at least two ways to increase the yield (amount) of ammonia based on this principle.
Answers: 2

Chemistry, 22.06.2019 09:00, dante766
Achemist 16 drop copper metal from copper chloride solution. the chemist place is 0.50 g of aluminum foil in a solution containing 0.75 g of copper (ii) chloride. a single replacement reaction takes place. which statement explains the maximum amount of copper that the chemist can extract using this reaction?
Answers: 1

Chemistry, 22.06.2019 11:50, bellojamilet410
What substance has a mass of 9.5g and volume of 2.1cm^3
Answers: 2

Chemistry, 22.06.2019 23:30, lizdeleon248
The sum of the oxidation numbers in a neutral compound is always
Answers: 2
Do you know the correct answer?
Carbon monoxide and chlorine combine in an equilibrium reaction to produce the highly toxic product,...
Questions in other subjects:

Biology, 17.07.2019 16:30

English, 17.07.2019 16:30



Mathematics, 17.07.2019 16:30

Biology, 17.07.2019 16:30


Social Studies, 17.07.2019 16:30

Chemistry, 17.07.2019 16:30

Biology, 17.07.2019 16:30


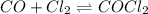
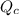 ) for this reaction is represented as:
) for this reaction is represented as:![Q_{c}=\frac{[COCl_{2}]}{[CO][Cl_{2}]}](/tpl/images/0619/3924/9faba.png)
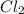 ] = 0.010 M and [
] = 0.010 M and [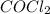 ] = 0.070 M
] = 0.070 M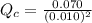 = 700
= 700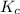 therefore reaction will proceed towards forward direction i.e. more
therefore reaction will proceed towards forward direction i.e. more



