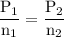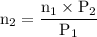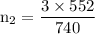Chemistry, 22.04.2020 03:59, davelopez979
A catalytic converter in an automobile uses a palladium or platinum catalyst to convert carbon monoxide gas to carbon dioxide according to the following reaction: 2CO(g) + O2 (g) --> 2CO2(g) A chemist researching the effectiveness of a new catalyst combines a 2.0 : 1.0 mole ratio mixture of carbon monoxide and oxygen gas (respectively) over the catalyst in a 2.45L flask at a total pressure of 740 torr and a temperature of 552 C . When the reaction is complete, the pressure in the flask has dropped to 552 torr. What percentage of the carbon monoxide was converted to carbon dioxide?

Answers: 1
Other questions on the subject: Chemistry

Chemistry, 21.06.2019 14:50, stodd9503
Your roll: experienced electron speech is adressed to: a new "freshman class" of electrons job: write a speech task: you are to pretend that you are giving a speech to a new group of electrons. be sure to mention their placement in an atom, their charge, and their role in chemical bonding (ionic and covalent) be specific!
Answers: 3



Chemistry, 22.06.2019 18:00, sandeebassett3
Mercury turns to vapor at 629.88 k how much heat is lost 175 g of mercury vapor at 650 current condenses to a liquid at 297 ca mercury turns to weber at 629.88 kelvin how much he is lost 175 g of mercury vapor and 650 coming condensers to liquidate 297 kevin
Answers: 2
Do you know the correct answer?
A catalytic converter in an automobile uses a palladium or platinum catalyst to convert carbon monox...
Questions in other subjects:

Mathematics, 04.10.2019 21:00


Mathematics, 04.10.2019 21:00



Mathematics, 04.10.2019 21:00




Mathematics, 04.10.2019 21:00


 2CO₂
2CO₂