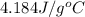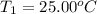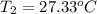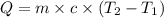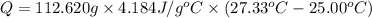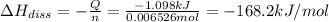Chemistry, 22.04.2020 03:29, milkshakegrande101
The salt magnesium chloride is soluble in water. When 0.620 g MgCl2 is dissolved in 112.00 g water, the temperature of the solution increases from 25.00 °C to 27.33 °C. Based on this observation, calculate the dissolution enthalpy, ΔdissH, of MgCl2. Assume that the specific heat capacity of the solution is 4.184 J g-1 °C-1 and that the energy transfer to the calorimeter is negligible. ΔdissH = kJ/mol

Answers: 1
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 02:30, ulilliareinhart2
The is a particle with one unit of positive charge a. proton b. positron c. electron d. nucleus awnser quick it is a important science test!
Answers: 2


Chemistry, 22.06.2019 05:30, jameskarbar9p8c9d2
Match the following vocabulary terms to their definitions. 1. amount of energy required to change 1 gram of material from the solid to the liquid state at its melting point 2. a measure of the kinetic energy of the particles of a substance 3. the amount of heat energy required to raise the temperature of 1 gram of liquid water from 14.5°c to 15.5°c 4. amount of energy required to change 1 gram of material from the liquid to the gaseous state at its boiling point 5. the amount of energy required to change 1 gram of a substance 1°c a. temperature b. latent heat of vaporization c. latent heat of fusion d. calorie e. specific heat
Answers: 1

Chemistry, 22.06.2019 10:30, perezanthony2403
Which describes fat? a: a carbohydrate that produces energy b: a nucleic acid that directs cell function c: a lipid that stores energy d: a protein that speeds up a chemical reaction
Answers: 1
Do you know the correct answer?
The salt magnesium chloride is soluble in water. When 0.620 g MgCl2 is dissolved in 112.00 g water,...
Questions in other subjects:




Mathematics, 30.11.2021 01:00

History, 30.11.2021 01:00

History, 30.11.2021 01:00


English, 30.11.2021 01:00