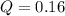Chemistry, 22.04.2020 02:39, LtotheJ0225
Nitric oxide, an important pollutant in air, is formed from the elements nitrogen and oxygen at high temperatures, such as those obtained when gasoline burns in an automobile engine. At 2000°C, K for the reaction N2(g) + O2(g) 2NO(g) is 0.01. Predict the direction in which the system will move to reach equilibrium at 2000°C if 0.4 moles of N 2, 0.1 moles of O 2, and 0.08 moles of NO are placed in a 1.0-liter container.

Answers: 1
Other questions on the subject: Chemistry



Do you know the correct answer?
Nitric oxide, an important pollutant in air, is formed from the elements nitrogen and oxygen at high...
Questions in other subjects:


Mathematics, 19.05.2020 15:58


Biology, 19.05.2020 15:58


Mathematics, 19.05.2020 15:58


Mathematics, 19.05.2020 15:58

Mathematics, 19.05.2020 15:58

Mathematics, 19.05.2020 15:58


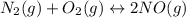
![Q=\frac{[NO]^2}{[N_2][O_2]}](/tpl/images/0617/1727/a703d.png)