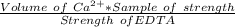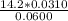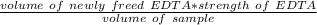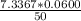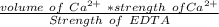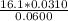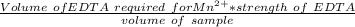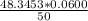Chemistry, 22.04.2020 01:05, cheeraly09
A 50.0 mL sample containing Cd2+ and Mn2+ was treated with 64.0 mL of 0.0600 M EDTA . Titration of the excess unreacted EDTA required 16.1 mL of 0.0310 M Ca2+ . The Cd2+ was displaced from EDTA by the addition of an excess of CN− . Titration of the newly freed EDTA required 14.2 mL of 0.0310 M Ca2+ . What are the concentrations of Cd2+ and Mn2+ in the original solution?

Answers: 2
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 06:00, momof7hardings
When would a bouncy ball have the most potential energy
Answers: 2

Chemistry, 22.06.2019 11:50, tajanaewilliams77
If oil spills continue, all of the following should be expected except (2 points) death of aquatic life. polluted groundwater. decreased soil productivity. increased global temperatures.
Answers: 3

Chemistry, 22.06.2019 12:30, fvmousdiana
In france, grapes are 1.95 euros per kilogram. what is the cost of grapes, in dollars per pound, if the exchange rate is 1.14 dollars/euro? (2.6)
Answers: 3

Chemistry, 22.06.2019 14:00, rosetoheart2
What mass of natural gas (ch4) must you burn to emit 276 kj of heat?
Answers: 2
Do you know the correct answer?
A 50.0 mL sample containing Cd2+ and Mn2+ was treated with 64.0 mL of 0.0600 M EDTA . Titration of t...
Questions in other subjects:

Mathematics, 17.10.2020 06:01

Biology, 17.10.2020 06:01

Biology, 17.10.2020 06:01

Arts, 17.10.2020 06:01

English, 17.10.2020 06:01






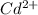 in the original solution= 0.0088 M
in the original solution= 0.0088 M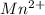 in the original solution = 0.058 M
in the original solution = 0.058 M