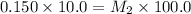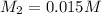Chemistry, 22.04.2020 00:04, sksksksksk1
A silver nitrate (AgNO3) solution is 0.150 M. 100.0 mL of a diluted silver nitrate solution is prepared using 10.0 mL of the more concentrated solution (which is then diluted with distilled water to 100.0 mL). Use the dilution equation to find the molarity of silver nitrate in the diluted solution.

Answers: 2
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 12:30, masteroftheuniverse3
When a scientific theory has been tested and proved by the scientific community, it becomes a law
Answers: 2


Chemistry, 22.06.2019 15:00, emmalie52
Phosphorous acid, h3po3(aq) , is a diprotic oxyacid that is an important compound in industry and agriculture. p k a1 p k a2 1.30 6.70 calculate the ph for each of the points in the titration of 50.0 ml of 1.5 m h3po3(aq) 1.5 m h 3 po 3 ( aq ) with 1.5 m koh(aq). 1.5 m koh ( aq ) .
Answers: 1

Chemistry, 22.06.2019 20:00, AaronEarlMerringer
What is the molar mass of the anhydrous compound? answer using four significant figures. 36.02 g/mol 120.15 g/mol 156.12 g/mol
Answers: 1
Do you know the correct answer?
A silver nitrate (AgNO3) solution is 0.150 M. 100.0 mL of a diluted silver nitrate solution is prepa...
Questions in other subjects:



Computers and Technology, 07.06.2021 04:40


English, 07.06.2021 04:40

Mathematics, 07.06.2021 04:40

Chemistry, 07.06.2021 04:40


Mathematics, 07.06.2021 04:40


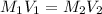
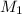 = molarity of concentrated solution = 0.150 M
= molarity of concentrated solution = 0.150 M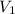 = volume of concentrated solution = 10.0 ml
= volume of concentrated solution = 10.0 ml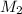 = molarity of diluted solution = ?
= molarity of diluted solution = ?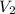 = volume of diluted solution = 100.0 ml
= volume of diluted solution = 100.0 ml