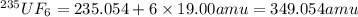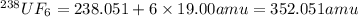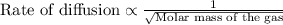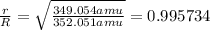Chemistry, 21.04.2020 20:49, elreemali03
We separate U-235 from U-238 by fluorinating a sample of uranium to form UF6 (which is a gas) and then taking advantage of the different rates of effusion and diffusion for compounds containing the two isotopes. Calculate the ratio of effusion rates for 238UF6 and 235UF6. The atomic mass of U-235 is 235.054 amu and that of U-238 is 238.051 amu.

Answers: 1
Other questions on the subject: Chemistry

Chemistry, 21.06.2019 19:50, limelight11
Which statement describes how phase changes can be diagrammed as a substance is heated? the phase is on the y-axis and the temperature is on the x-axis. the temperature is on the y-axis and the phase is on the x-axis. the time is on the y-axis and the temperature is on the x-axis. the temperature is on the y-axis and the time is on the x-axis.
Answers: 1

Chemistry, 22.06.2019 09:00, bibhu42kumarp7o4ss
At 300 mm hg, a gas has a volume of 380 l, what is the volume at standard pressure
Answers: 1

Chemistry, 22.06.2019 10:00, melissa9882
A50.0g sample of liquid water at 0.0 c ends up as ice at -20.0 c. how much energy is involved in this change?
Answers: 1
Do you know the correct answer?
We separate U-235 from U-238 by fluorinating a sample of uranium to form UF6 (which is a gas) and th...
Questions in other subjects:

English, 06.05.2020 06:38




Mathematics, 06.05.2020 06:38



Mathematics, 06.05.2020 06:38

Mathematics, 06.05.2020 06:38


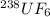 and
and 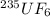 is 0.995734.
is 0.995734.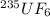 gas =
gas = 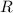
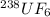 gas =
gas =