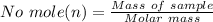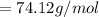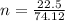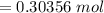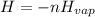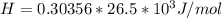Chemistry, 21.04.2020 18:07, roseemariehunter12
The following information is given for ether, C2H5OC2H5, at 1atm: boiling point = 34.6 °C Hvap(34.6 °C) = 26.5 kJ/mol specific heat liquid = 2.32 J/g°C /At a pressure of 1 atm, what is H in kJ for the process of condensing a 22.5 g sample of gaseous ether at its normal boiling point of 34.6 °C.

Answers: 1
Other questions on the subject: Chemistry

Chemistry, 21.06.2019 19:30, angelinararr5783
Which answer lists the fundamental forces in order from strongest to weakest
Answers: 1

Chemistry, 22.06.2019 06:30, cadenhuggins2
Predict whether the changes in enthalpy, entropy, and free energy will be positive or negative for the boiling of water, and explain your predictions. how does temperature affect the spontaneity of this process?
Answers: 1

Chemistry, 22.06.2019 11:00, blondieb1722
Which are examples of how technology has advanced scientific understanding.1using hot water to sterilize medical equipment.2transplanting a human organ into another individual.3inserting genes from one sheep into another cell to make a cloneunderstanding the different structures that make up a cell.4examining microorganisms from the deepest parts of the ocean
Answers: 2

Chemistry, 22.06.2019 12:00, KKHeffner02
Which statement best explains the relationship between an area is geography and the temperature of its surface water
Answers: 1
Do you know the correct answer?
The following information is given for ether, C2H5OC2H5, at 1atm: boiling point = 34.6 °C Hvap(34.6...
Questions in other subjects:

Mathematics, 17.12.2020 19:30

Mathematics, 17.12.2020 19:30

History, 17.12.2020 19:30


Social Studies, 17.12.2020 19:30

Mathematics, 17.12.2020 19:30

English, 17.12.2020 19:30

Mathematics, 17.12.2020 19:30


Social Studies, 17.12.2020 19:30


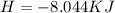
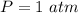
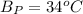
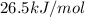

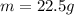
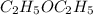 is given as
is given as