Chemistry, 21.04.2020 17:42, etaylor3421
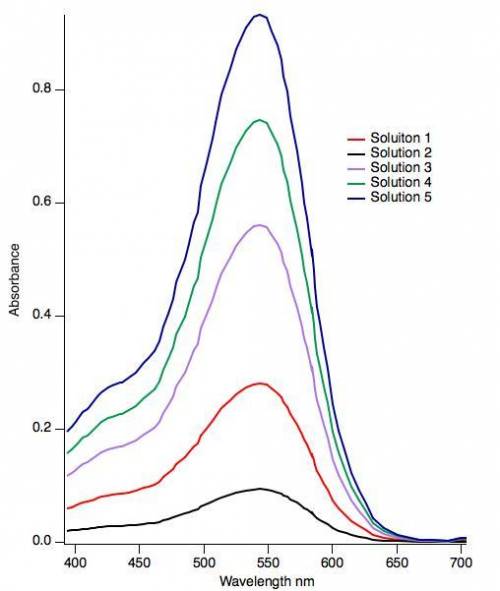
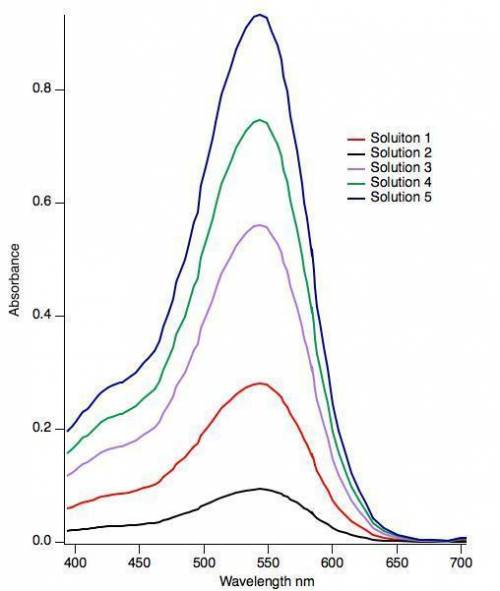
The absorbance features observed in the visible spectrum for curcumin is a result of an allowed optical excitation of an electron from the π-HOMO to the π*-LUMO. What is the energy change for this electronic excitation based on the spectrum? (Hint: Energy and wavelength are related by the equation E = hc/λ.) h = 4.136 × 10-15 eV⋅ s c = 2.998 × 108 m/s

Answers: 1
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 03:40, jude3412
In an effort to address concerns about global warming, a power plant in portland, oregon is designed to take all of its exhaust gases from its boilers and recycle the co2 using the solvay process to make sodium hydrogen carbonate. the reaction is shown below. nh3(g) + h2o(l) + co2(g) + nacl(aq) → nahco3(aq) + nh4cl(aq) how many liters each of nh3 and co2 (both at stp) would be consumed to produce 3.00 kg of sodium bicarbonate? the volume of both nh3 and co2 would be
Answers: 1

Chemistry, 22.06.2019 05:30, jzjajsbdb8035
Which other elements contain the same number of outer electrons as sodium
Answers: 3

Chemistry, 22.06.2019 13:00, cnfndbxbfbdb2031
Which of the following are good traits of a hypothesis? it will be able to be testedit can predict an outcomeit will explain the observationsall of these
Answers: 2
Do you know the correct answer?
The absorbance features observed in the visible spectrum for curcumin is a result of an allowed opti...
Questions in other subjects:

Mathematics, 13.01.2021 04:00


English, 13.01.2021 04:00

Mathematics, 13.01.2021 04:00

Mathematics, 13.01.2021 04:00

Geography, 13.01.2021 04:00