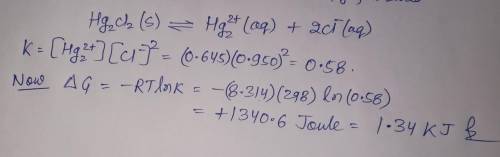Chemistry, 21.04.2020 04:51, micahwilkerson9495
A chemist fills a reaction vessel with 0.623g mercurous chloride(Hg2Cl2) solid, 0.645M mercury (I) (Hg2^2+)aqueous solution, and 0.905M chloride (Cl-) aqueous solution at a temperature of 25.0°C.
Under these conditions, calculate the reaction free energy ΔG for the following chemical reaction:
Hg2Cl2(s) ⇌ Hg2^2+ (aq) + 2Cl^- (aq)

Answers: 1
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 06:30, irvinbhangal2
What effect might melting sea ice have for people who live in coastal areas?
Answers: 1

Chemistry, 22.06.2019 12:30, MrSavannahCat
Clyde and marilyn are riding a roller coaster. during which section(s) of the track is their potential energy converted to kinetic energy? a. from point b to point c only b. from point b to point d only c. from point a to point b only d. from point a to point b and from point c to point d
Answers: 1


Chemistry, 23.06.2019 02:00, hayleebeals50
To calculate the molarity of a solution, you need to know the moles of solute and the
Answers: 2
Do you know the correct answer?
A chemist fills a reaction vessel with 0.623g mercurous chloride(Hg2Cl2) solid, 0.645M mercury (I) (...
Questions in other subjects:

Mathematics, 05.11.2020 16:40




Mathematics, 05.11.2020 16:40