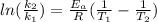The rate constant for a first order reaction is 0.060s-1. when the temperature is increased from 298K to 331 K , the rate constant increases to 0.18s-1. Calculate the activation energy for this reaction. Use kJ/mol for the units. The activation energy in kJ/mol equals (include the units in your answer):

Answers: 1
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 07:00, mayamabjishovrvq9
The variability in marine salinity between habitats does not impact the fish living there. select the best answer from the choices provided t f
Answers: 1


Chemistry, 22.06.2019 23:10, RealStephani
Match the formula for the following compound: magnesium sulfate heptahydratemgs·7h2omg2so4·7h2omg(s o4)2·7h2omgso4·7h2o
Answers: 1

Chemistry, 23.06.2019 01:30, heavendl13
How is the solubility of a carbon dioxide gas in water increase?
Answers: 1
Do you know the correct answer?
The rate constant for a first order reaction is 0.060s-1. when the temperature is increased from 298...
Questions in other subjects:

Mathematics, 27.06.2019 15:00

English, 27.06.2019 15:00

Mathematics, 27.06.2019 15:00


History, 27.06.2019 15:00



Physics, 27.06.2019 15:00

Mathematics, 27.06.2019 15:00