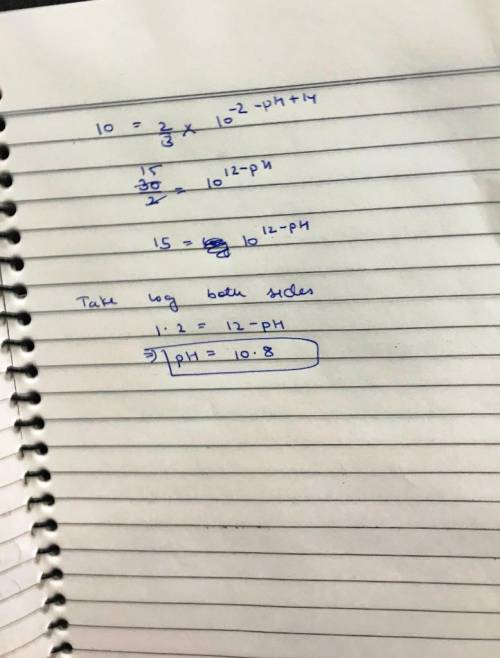Chemistry, 16.04.2020 00:17, sarahhN7534
The decomposition of Bromodichloroacetate BrCl2CCO2- is an important required step in water purification. The kinetics of such decomposition has been presented in Chemical Reviews, November 2001. There are two possible pathways for these reactions, one unimolecular and the other bimolecular with the help of OH- ions.
Path 1. BrCl2CCO2- + H2O goes to CHCl2Br + HCO3-
With a pseudo-first-order rate constant k1=1.6 x 10-6 1/sec
Path 2. BrCl2CCO2- + OH- goes to Cl2OHCCO2- + Br-
With a second-order rate constant k2=2.4x10-4 1/(M sec)
(a) Write the overall rate law for the disappearance of BrCl2CCO2.
(b) What is the ratio of the rates for paths 1 and 2 at pH=7?
(c) At what pH would the rate for path 2 be 10% of the rate for path 1?

Answers: 3
Other questions on the subject: Chemistry


Chemistry, 22.06.2019 03:50, daniel9299
Consider the reaction: n2(g) + o2(g) ? 2no(g) kc = 0.10 at 2000oc starting with initial concentrations of 0.040 mol/l of n2 and 0.040 mol/l of o2, calculate the equilibrium concentration of no in mol/l how would this be done?
Answers: 3

Chemistry, 22.06.2019 09:00, SilverTheAmarok
This chart lists four kinds of polymers and their sources. what can be known about all four polymers, despite their differences? they come from living things. they share ionic carbon bonds. they are at least 100 monomers long. they are made of repeating subunits.
Answers: 2

Chemistry, 22.06.2019 23:00, hailey5campbelp7d1c0
In which region is the substance in both the solid phase and the liquid phase? 1 2. 3 4 mark this and return save and exit next
Answers: 2
Do you know the correct answer?
The decomposition of Bromodichloroacetate BrCl2CCO2- is an important required step in water purifica...
Questions in other subjects:

Mathematics, 15.09.2020 01:01

Mathematics, 15.09.2020 01:01

Chemistry, 15.09.2020 01:01

English, 15.09.2020 01:01

Social Studies, 15.09.2020 01:01

Mathematics, 15.09.2020 01:01

Geography, 15.09.2020 01:01

Mathematics, 15.09.2020 01:01

Mathematics, 15.09.2020 01:01

Mathematics, 15.09.2020 01:01