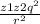GThe atomic radii of a divalent cation and a monovalent anion are 0.085 nm and 0.125 nm, respectively. (a) Calculate the force of attraction between these two ions at their equilibrium interionic separation (i. e., when the ions just touch one another)./homework-help/electron-su bshell-filled-rare-earth-series-ele ments-periodic-chapter-2-problem-14 qp-solution-9781118324578-exc

Answers: 1
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 15:40, alleshia2007
Use the periodic table to complete this equation that represents nuclear fission processesun - ba c 3 n
Answers: 2

Chemistry, 22.06.2019 17:10, sophiaa23
)benzene and toluene form nearly ideal solutions. consider an equimolar solution of benzene and toluene. at 20 °c the vapour pressures of pure benzene and toluene are 9.9 kpa and 2.9 kpa, respectively. the solution is boiled by reducing the external pressure below the vapour pressure. calculate (i) the pressure when boiling begins, (ii) the composition of each component in the vapour, and (iii) the vapour pressure when only a few drops of liquid remain. assume that the rate of vaporization is low enough for the temperature to remain constant at 20 °c.
Answers: 1

Chemistry, 22.06.2019 22:10, preachersgirl5
What is the indicator of the number of ions in solution? the amount of conductivity the amount of precipitate the amount of solute added
Answers: 1

Do you know the correct answer?
GThe atomic radii of a divalent cation and a monovalent anion are 0.085 nm and 0.125 nm, respectivel...
Questions in other subjects:

Biology, 18.06.2021 03:00


Mathematics, 18.06.2021 03:00

Mathematics, 18.06.2021 03:00


Mathematics, 18.06.2021 03:00

Mathematics, 18.06.2021 03:00

Chemistry, 18.06.2021 03:00



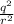 = k
= k