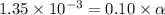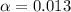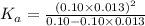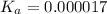Chemistry, 15.04.2020 20:21, tiffanybrandy23
Determine the acid dissociation constant for a 0.10 m acetic acid solution that has a ph of 2.87. Acetic acid is a weak monoprotic acid and the equilibrium equation of interest is

Answers: 1
Other questions on the subject: Chemistry

Chemistry, 21.06.2019 17:30, roseemariehunter12
When the earth was formed and cooled, why did nickel and iron end up in the center of the earth while basalt and granite ended up in the outer layers
Answers: 3

Chemistry, 21.06.2019 21:40, taysomoneyyy
During trial 2, what allowed you to determine that aluminum was the limiting reactant? check all that apply. all of the copper dissolved. all of the aluminum dissolved. the solution turned clear. the number of grams of copper(ii) chloride used in the reaction was greater than the number of grams of aluminum. the molar ratio of copper(ii) chloride to aluminum was greater than 3: 2, the equation’s molar ratio.
Answers: 2

Chemistry, 22.06.2019 02:50, Jerrikasmith28
The conventional equilibrium constant expression (kc) for the system below is: 2icl(s) ⇄ i2(s) + cl2(g) [cl2] ([i2] + [cl2])/2[icl] [i2][cl2]/[icl]2 none of the listed answers are correct [i2][cl2]/2[icl]
Answers: 2
Do you know the correct answer?
Determine the acid dissociation constant for a 0.10 m acetic acid solution that has a ph of 2.87. Ac...
Questions in other subjects:



Mathematics, 21.06.2019 13:00


Mathematics, 21.06.2019 13:00


Mathematics, 21.06.2019 13:00

History, 21.06.2019 13:00

Mathematics, 21.06.2019 13:00

Biology, 21.06.2019 13:00


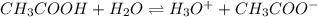
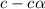
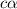
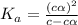
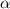 = dissociation constant = ?
= dissociation constant = ?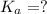
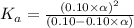
![pH=-log[H^+]](/tpl/images/0602/6192/15713.png)
![2.87=-log[H^+]](/tpl/images/0602/6192/3a07c.png)
![[H^+]=1.35\times 10^{-3}](/tpl/images/0602/6192/8f9f0.png)
![[H^+]=c\times \alpha](/tpl/images/0602/6192/4fc41.png)