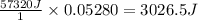A student enters the lab and conducts Part A of the Experiment. The student uses 25.00 mL of 2.112 M HCl, and adds NaOH in excess as instructed. If the ΔH of the neutralization reaction is known to be -57,320 J/mol H2O, what is the total theoretical heat released (in Joules)?

Answers: 1
Other questions on the subject: Chemistry

Chemistry, 21.06.2019 13:10, puchie1225
Cobalt-60 is an artificial radioisotope that is produced in a nuclear reactor and is used as a gamma-ray source in the treatment of certain types of cancer. if the wavelength of the gamma radiation from a cobalt-60 source is 1.00 × 10-3 nm, calculate the energy of a photon of this radiation.
Answers: 2

Chemistry, 21.06.2019 17:10, codeyhatch142
Nitric oxide (no) can be formed from nitrogen, hydrogen and oxygen in two steps. in the first step, nitrogen and hydrogen react to form ammonia: n2(g) + 2 h_2(g) rightarrow 2 nh_3 (g) delta h = -92. kj in the second step, ammonia and oxygen react to form nitric oxide and water: 4 nh_3(g) + 5 o_2(g) rightarrow 4no(g) + 6 h_2o(g) delta h = -905. kj calculate the net change in enthalpy for the formation of one mole of nitric oxide from nitrogen, hydrogen and oxygen from these reactions. round your answer to the nearest .
Answers: 1
Do you know the correct answer?
A student enters the lab and conducts Part A of the Experiment. The student uses 25.00 mL of 2.112 M...
Questions in other subjects:



Chemistry, 14.07.2020 01:01

Mathematics, 14.07.2020 01:01

Spanish, 14.07.2020 01:01




Mathematics, 14.07.2020 01:01

Physics, 14.07.2020 01:01


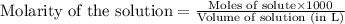 .....(1)
.....(1)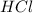 solution = 2.112 M
solution = 2.112 M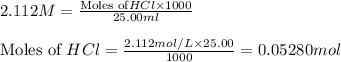

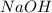 is the excess reagent.
is the excess reagent.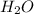
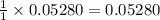 moles of
moles of