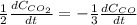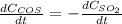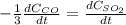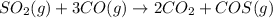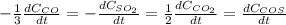Chemistry, 15.04.2020 17:39, tamariarodrigiez
Sulfur dioxide emissions in power plant stack gases may react with carbon monoxide as follows: SO_2 (g) + 3CO(g) rightarrow 2CO_2 + COS(g) Write an equation relating each of the following pairs of rates:
a. The rate of formation of CO_2 to the rate of consumption of CO
b. The rate of formation of COS to the rate of consumption of SO_2
c. The rate of consumption of CO to the rate of consumption of SO_2

Answers: 3
Other questions on the subject: Chemistry


Chemistry, 22.06.2019 10:10, ragegamer334p3xlso
Stage in which a star’s outer layers have started to cool and grow outward?
Answers: 3

Chemistry, 23.06.2019 00:30, vane6176
You are attempting to recrystallize a crude product mixture. you add the appropriate amount of hot solvent and are allowing the solution to slowly cool to room temperature. however, at room temperature no crystals have appeared, which of the following methods should be used to induce crystallization? choose all correct answers. a) place the flask in an ice bath. b) swirl the contents of the flask. c) add a small seed crystal of the desired product. d) scratch the inside of the glassware using a stir rod. it can be multiple choices
Answers: 3
Do you know the correct answer?
Sulfur dioxide emissions in power plant stack gases may react with carbon monoxide as follows: SO_2...
Questions in other subjects:





Mathematics, 24.08.2021 03:10




Mathematics, 24.08.2021 03:10

Mathematics, 24.08.2021 03:10