Chemistry, 15.04.2020 15:52, crystalclear99
A chemistry student weighs out 0.09666 g of phosphoric acid (H3PO4), a triprotic acid, into a 250.volumetric flask and dilutes to the mark with distilled water. He plans to titrate the acid with 0.2000 M NaoH solution. Calculate the volume of NaoH solution the student will need to add to reach the final equivalence point. Round your answer to 3 significant digits.

Answers: 2
Other questions on the subject: Chemistry


Chemistry, 22.06.2019 22:30, skinniestoflegends
Consider a culture medium on which only gram-positive organisms such as staphylococcus aureus colonies can grow due to an elevated nacl level. a yellow halo surrounds the growth, indicating the bacterium fermented a sugar in the medium, decreasing the ph as a result and changing the color of a ph indicator chemical. this type of medium would be referred to as a differential and enrichment culture.
Answers: 2

Do you know the correct answer?
A chemistry student weighs out 0.09666 g of phosphoric acid (H3PO4), a triprotic acid, into a 250.vo...
Questions in other subjects:


Mathematics, 30.10.2020 16:50

Mathematics, 30.10.2020 16:50



Mathematics, 30.10.2020 16:50





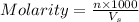
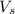 = volume of solution in ml = 250 ml
= volume of solution in ml = 250 ml


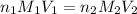
 are the n-factor, molarity and volume of acid which is
are the n-factor, molarity and volume of acid which is 
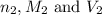 are the n-factor, molarity and volume of base which is NaOH.
are the n-factor, molarity and volume of base which is NaOH.