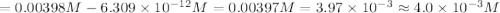Chemistry, 18.10.2019 22:30, cmfuentes0816
The equation for the ph of a substance is ph = –log[h+], where h+ is the concentration of hydrogen ions. a basic solution has a ph of 11.2. an acidic solution has a ph of 2.4. what is the approximate difference in the concentration of hydrogen ions between the two solutions? a) 1.6*10^-9 b) 4.0*10^-3 c)6.7*10^-1 d)1.6*10^-1

Answers: 2
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 04:40, khan2491
Silver tarnishes as silver metal reacts with hydrogen sulfide, h2s, in the air. in this reaction, dark silver sulfide, au2s, covers the surface of silver. when silver is polished, this coating of silver sulfide can be removed from the surface. this makes the silver shiny again. enter the coefficients that balance the tarnishing reaction equation. (type 1 for no coefficient.)
Answers: 2


Chemistry, 22.06.2019 13:50, aesthetickait
How does the motion of particles in a gas change as the gas cools
Answers: 2

Do you know the correct answer?
The equation for the ph of a substance is ph = –log[h+], where h+ is the concentration of hydrogen i...
Questions in other subjects:


Mathematics, 06.04.2021 18:20

Mathematics, 06.04.2021 18:20




Mathematics, 06.04.2021 18:20




![pH = -\log[H+]](/tpl/images/0332/6029/30b50.png)
![11.2=-\log[H^+]](/tpl/images/0332/6029/fa6ad.png)
![[H^+]=6.309\times 10^{-12} M](/tpl/images/0332/6029/eb264.png)
![2.4=-\log[H^+]](/tpl/images/0332/6029/156c9.png)
![[H^+]'=0.0039810 M=3.9810\times 10^{-3} M](/tpl/images/0332/6029/c1356.png)
![=[H^+]'-[H^+]](/tpl/images/0332/6029/74fcc.png)