
A dive tank is designed and built to be safe if the difference between the pressure inside the tank and the pressure outside the tank does not exceed 12 atm. A diver has the tank filled at the dive store at a temperature of 20o C to a pressure of 10 atm. The diver places the tank into his car and parks the car in the sun where the temperature of the gas in the tank increases to 60o C. Is the tank safe at this temperature? You must show the setup of an equation, the final answer with units , and a short statement whether the tank is safe or not.

Answers: 1
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 16:00, sassy11111515
The chemical equation below shows the reaction of sodium (na) and chlorine (cl) to form sodium chloride (nacl). 2na + cl2 → 2nacl in this equation, which of the following is a reactant? i. sodium ii. chlorine iii. sodium chloride
Answers: 1

Chemistry, 22.06.2019 19:00, Jasoncookies23
How does kepler second law of planetary motion overthrow one of the basic beliefs of classical astronomy
Answers: 1

Chemistry, 22.06.2019 19:30, simihehe
Phosphorous can form an ion called phosphide, which has the formula p3−. this ion can form an ion called phosphide, which has the formula p3−. this ion properties very similar to those of pforms when a phosphorus atom loses three protonsis called a cationcontains 18 electrons
Answers: 2
Do you know the correct answer?
A dive tank is designed and built to be safe if the difference between the pressure inside the tank...
Questions in other subjects:

World Languages, 08.03.2021 19:40

Mathematics, 08.03.2021 19:40










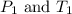 are the initial pressure and temperature of the gas.
are the initial pressure and temperature of the gas.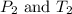 are the final pressure and temperature of the gas.
are the final pressure and temperature of the gas.





