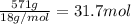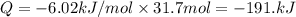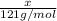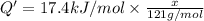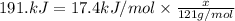A particular refrigerator cools by evaporating liquefied dichlorodifluoromethane, CCl2F2. How many kilograms of this liquid must be evaporated to freeze a tray of water at 0°C to ice at 0°C? The mass of the water is 571 g, the heat of fusion of ice is 6.02 kJ/mol, and the heat of vaporization of dichlorodifluoromethane is 17.4 kJ/mol.

Answers: 2
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 02:30, Countryqueen525
Asa choose the correct set of reaction coefficients to properly balance the following chemical equation according to the law of conservation of mass: __s8 + __o2 ==> __so2 1, 1, 8 1, 8, 1 1, 8, 8 8, 1, 1
Answers: 1

Chemistry, 22.06.2019 20:30, sydneip6174
We are hoping to create 5.72 grams of glucose. the plant was given 4.75 liters of co2 and 2.81 g of h20. which reactant was the limiting reagent? how much excess mass did we have of the other reactant?
Answers: 2

Chemistry, 22.06.2019 21:00, andrethisman88
Kp is the equilibrium constant for dissociation of the propionic acid dimer. what is the sign of the slope for a plot of the natural logarithm of kp vs. inverse temperature for this reaction?
Answers: 1

Chemistry, 22.06.2019 22:30, creepycrepes
Which of the following is not an assumption that scientists must make about the natural world? a. regularity b. causality c. predictability d. plausibility
Answers: 1
Do you know the correct answer?
A particular refrigerator cools by evaporating liquefied dichlorodifluoromethane, CCl2F2. How many k...
Questions in other subjects:

Mathematics, 04.10.2019 18:30


Spanish, 04.10.2019 18:30


Social Studies, 04.10.2019 18:30

English, 04.10.2019 18:30

History, 04.10.2019 18:30

Mathematics, 04.10.2019 18:30

Biology, 04.10.2019 18:30