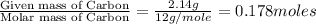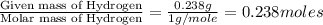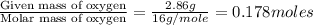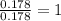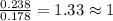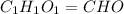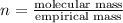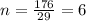Chemistry, 14.04.2020 20:14, kalebbenton15
Ascorbic acid (vitamin C) contains C, H, and O. In one combustion analysis, 5.24 g of ascorbic acid yields 7.86 g CO2 and 2.14 g H2O. Calculate the empirical formula and molecular formula of ascorbic acid given that its molar mass is about 176 g.

Answers: 2
Other questions on the subject: Chemistry

Chemistry, 21.06.2019 14:20, Mordred9571
Which is true of chemicals? a. things containing chemicals always cost a lot of money. b. chemicals are never dangerous. c. chemicals are in many substances in a home. d. chemicals are rarely found on earth.
Answers: 1

Chemistry, 21.06.2019 18:30, mamasmontoya
For each of the following mixtures decide if filtering would be suitable to separate the substances. explain your answers. oil in water sugar in water sand in water chalk in water tea leaves in a cup of tea
Answers: 2

Do you know the correct answer?
Ascorbic acid (vitamin C) contains C, H, and O. In one combustion analysis, 5.24 g of ascorbic acid...
Questions in other subjects:


Mathematics, 27.10.2020 20:10


English, 27.10.2020 20:10





Mathematics, 27.10.2020 20:10


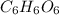
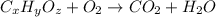
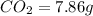
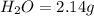
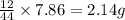 of carbon will be contained.
of carbon will be contained.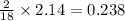 of hydrogen will be contained.
of hydrogen will be contained.