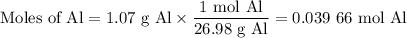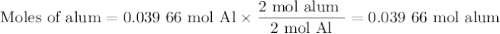Chemistry, 14.04.2020 18:53, sierravick123owr441
Scaled Synthesis of Alum. Show your calculations for:a. the experimental scaling factor giving rise to a 15.0 g theoretical yield;b. the corrected volumes of KOH and H2SO4; andc. the theoretical yield of alum based on the actual amount of Al used. Make sure you carefully show each step for these calculations.

Answers: 2
Other questions on the subject: Chemistry


Chemistry, 22.06.2019 05:50, vanessa051266
In an exothermic reaction the bonding energy of the product is: less than the reactants same as the reactants greater than the reactants dependent upon the presence of a catalyst
Answers: 1

Chemistry, 22.06.2019 13:50, kelonmazon2492
Read the chemical equation. 2c2h2 + 5o2 → 4co2 + 2h2o which of the following statements would be correct if one mole of c2h2 was used in this reaction? one mole of oxygen was used in this reaction. five moles of oxygen were used in this reaction. four moles of carbon dioxide were produced from this reaction. two moles of carbon dioxide were produced from this reaction.
Answers: 3

Chemistry, 22.06.2019 16:00, graciewyatt6833
Sulfuric acid is a polyprotic acid. write balanced chemical equations for the sequence of reactions that sulfuric acid can undergo when it's dissolved in water.
Answers: 2
Do you know the correct answer?
Scaled Synthesis of Alum. Show your calculations for:a. the experimental scaling factor giving rise...
Questions in other subjects:






Physics, 26.06.2019 07:30

Physics, 26.06.2019 07:30