Chemistry, 13.04.2020 23:20, NicoleQuinn
An exhaled air bubble underwater at 290.
kPa has volume 18.9mL. What will be its
volume at the water's surface where the
pressure is 104kPa. (Why must a diver
exhale during ascent?)

Answers: 3
Other questions on the subject: Chemistry


Chemistry, 22.06.2019 16:00, hjgjlgkjg
Inside a flashbulb, oxygen surrounds a thin coil of magnesium. when the flashbulb is set off, a chemical reaction takes place in which magnesium combines with oxygen to form magnesium oxide. which of the chemical equations matches the reaction above? a. mg + o2 mgo2 + energy b. 2mg + o mg2o + energy c. 2mg + o2 2mgo + energy d. mg + o mgo + energy
Answers: 1

Chemistry, 22.06.2019 17:10, glitterpanda2468
Calculate the estimated density of each ball. use the formula d = m/v where d is the density, m is the mass, and v is the volume. record your calculations in table a of your student guide. given that the density of water is 1.0 g/cm3, make a prediction about whether each ball will float in water. record your prediction in table a. what is the estimated density of the table tennis ball? record your answer to the nearest hundredth
Answers: 2

Chemistry, 22.06.2019 17:30, Naysa150724
Oil rich countries in the middle east cover about 4% of earths total land area but prossess about 48% of the worlds known oil reserves what is the main reason for high concentration of reserves in this part of the world
Answers: 3
Do you know the correct answer?
An exhaled air bubble underwater at 290.
kPa has volume 18.9mL. What will be its
volume...
kPa has volume 18.9mL. What will be its
volume...
Questions in other subjects:

Computers and Technology, 31.01.2020 03:51

Mathematics, 31.01.2020 03:51

Social Studies, 31.01.2020 03:51

Mathematics, 31.01.2020 03:51



Geography, 31.01.2020 03:51

Spanish, 31.01.2020 03:51

Biology, 31.01.2020 03:51

Mathematics, 31.01.2020 03:51


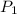 ) = 290 kPa
) = 290 kPa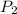 ) = 104 kPa
) = 104 kPa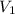 ) = 18.9 ml
) = 18.9 ml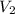 )
)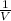
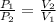
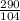 =
=