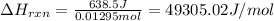Chemistry, 31.01.2020 02:45, rileyeddins1010
The addition of hydrochloric acid to a silver nitrate solution precipitates silver chloride according to the reaction: agno3(aq)+hcl(aq)→agcl(s)+hno3(aq). when you combine 70.0ml of 0.185m agno3 with 70.0ml of 0.185m hcl in a coffee-cup calorimeter, the temperature changes from 23.16∘c to 24.25∘c.. calculate δhrxn for the reaction as written. use 1.00 g/ml as the density of the solution and c=4.18j/g⋅∘c as the specific heat capacity.

Answers: 1
Other questions on the subject: Chemistry

Chemistry, 21.06.2019 16:30, monithebtslover01
Find the empirical formula of each of the following compounds. given mass or for each element in a sample of the compound 3,611 g ca; 6.389 g c1
Answers: 1


Chemistry, 22.06.2019 13:00, devontemiles8868
One of the hopes for solving the world's energy problem is to make use of the fusion reaction 21h +31h --> 42he + 10n + energy how much energy is released when 1 mol of deuterium is fused with 1 mol of tritium according to the above reaction? the masses of the atoms and the neutrons are as follows: 21h = 2.0140 amu 31h = 3.01605 amu 42he = 4.002603 amu 10n = 1.008665 amu. the speed of light is 2.9979 x 108 m/s.
Answers: 1

Chemistry, 23.06.2019 00:00, PineappleDevil889
2-bromo-2-methylbutane undergoes an e1 elimination reaction in the presence of ethanol. in the next reaction only one of the possible products is represented. although the product shown is not the major product of the reaction, notice that there is more than one way it can be produced. complete the mechanism and draw the missing substances.
Answers: 1
Do you know the correct answer?
The addition of hydrochloric acid to a silver nitrate solution precipitates silver chloride accordin...
Questions in other subjects:


English, 19.03.2021 21:40

Mathematics, 19.03.2021 21:40




Mathematics, 19.03.2021 21:40




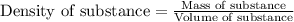
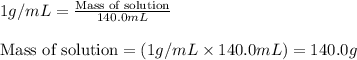
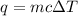
 = change in temperature =
= change in temperature = ![[24.25-23.16]^oC=1.09^oC](/tpl/images/0488/1311/2253a.png)
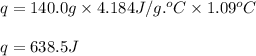
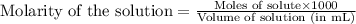
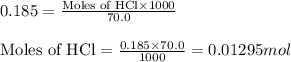
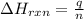
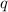 = amount of heat absorbed = 638.5 J
= amount of heat absorbed = 638.5 J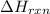 = enthalpy change of the reaction
= enthalpy change of the reaction