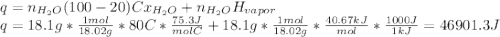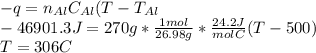Chemistry, 11.04.2020 04:49, goreeefk4939
18.1 g of water (initially at 20 oC) is poured onto 270 g of hot, aluminum metal (initially at 500 oC). Once all of the water has vaporized (and no heat is lost to the surrounding air) what will be the final temperature of the aluminum?
a. MPAl = 660 oC
b. Csolid Al = 24.2 J/mol oC
c. Cliquid Al = 29.3 J/mol oC
d. Cliquid water = 75.3 J/mol oC
e. Cwater vapor = 33.1 J/mol oC
f.ΔHfus water = 6.02 kJ/mol
g.ΔHvap water = 40.67 kJ/mol

Answers: 3
Other questions on the subject: Chemistry


Chemistry, 22.06.2019 10:30, Clivensp5
Skills of homo sapiens were found an excavation. the skulls were preserved because the bodies were frozen. so, these fossils are (blank) fossils. the image shows the evolution of skulls beginning 2 to 3 million years ago. based on the image, modern human skulls(blank) ape skulls.
Answers: 1

Chemistry, 22.06.2019 15:10, strodersage
Which statement describes the phase change that occurs when dry ice is placed in an open container at room temperature?
Answers: 1

Chemistry, 22.06.2019 20:40, ohgeezy
Select the correct value for the indicated bond angle in each of the compounds. o−o−oo−o−o angle of o3 90° 109.5° < 109.5° 120° < 120° 180° f−b−ff−b−f angle of bf3 180° < 109.5° < 120° 120° 109.5° 90° f−o−ff−o−f angle of of2 < 120° 120° 90° 109.5° 180° < 109.5° cl−be−clcl−be−cl angle of becl2 90° 109.5° 180° 120° < 109.5° < 120° f−p−ff−p−f angle of pf3 90° 109.5° < 109.5° 180° 120° < 120° h−c−hh−c−h angle of ch4 90° < 109.5° 180° 120° < 120° 109.5°
Answers: 1
Do you know the correct answer?
18.1 g of water (initially at 20 oC) is poured onto 270 g of hot, aluminum metal (initially at 500 o...
Questions in other subjects:




Mathematics, 05.09.2020 18:01

English, 05.09.2020 18:01