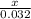Lily is preparing two samples different
compounds so that each sample contains the
same mass of calcium. The first sample she
prepares is 10.0 grams of calcium phosphate,
Ca3(PO4)2. She is preparing her second sample
using calcium hydroxide, Ca(OH)2. What mass of
Ca(OH)2 should she prepare so that it contains
the same mass of calcium as the first sample?
Help?

Answers: 3
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 06:00, palomaresmitchelle
There are 6.022, 104 atoms of hg in 1 mole of hg the number of atoms in 45 moles of hg can be found by multiplying 4.5 by 6.022, 102 which is the number of atoms in 4.5 moles of hg, correctly written in scientific notation with the correct number of significant figures? 0 21,109 0 21,100 271, 1024 27.099, 100 mark this and retum save and exit submit
Answers: 1

Chemistry, 22.06.2019 11:00, daniel1480
Problem page combustion of hydrocarbons such as pentane ( c5 h12 ) produces carbon dioxide, a "greenhouse gas." greenhouse gases in the earth's atmosphere can trap the sun's heat, raising the average temperature of the earth. for this reason there has been a great deal of international discussion about whether to regulate the production of carbon dioxide.(a) write a balanced chemical equation, including physical state symbols, for the combustion of liquid pentane into gaseous carbon dioxide and gaseous water. (b) suppose 0.350 kg of pentane are burned in air at a pressure of exactly 1 atm and a temperature of 20.0 degree c. calculate the volume of carbon dioxide gas that is produced. be sure your answer has the correct number of significant digits.
Answers: 2

Do you know the correct answer?
Lily is preparing two samples different
compounds so that each sample contains the
same...
compounds so that each sample contains the
same...
Questions in other subjects:


Social Studies, 15.10.2019 03:00


English, 15.10.2019 03:00






Mathematics, 15.10.2019 03:00


 =
=