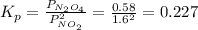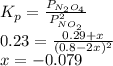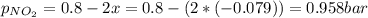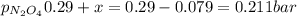N2O4 decomposes into NO2. At a certain temperature, the equilibrium pressures of NO2 and N2O4 are 1.6 bar and 0.58 bar, respectively. If the volume of the container is doubled at constant temperature, what would be the partial pressures of the gases when equilibrium is re-established

Answers: 1
Other questions on the subject: Chemistry

Chemistry, 21.06.2019 18:30, ciarakelly636owuiup
Asample of the male sex hormone testosterone, c19h28o2, contains 3.88×10^21 atoms of hydrogen.(a) how many atoms of carbon does it contain? (b) how many molecules of testosterone does it contain? (c) how many moles of testosterone does it contain? (d) what is the mass of this sample in grams?
Answers: 1

Chemistry, 22.06.2019 00:30, natalie1755
Butadiene undergoes a reaction at a certain temperature in the gas phase as follows: 2c4h6(g) --> c8h12(g) the following data were collected for this reaction: time (min) [c4h6] (m) 0 0.36 15 0.30 30 0.25 48 0.19 75 0. determine the order of the reaction and the rate constant. 1st order and k = 4.3x10 -4 s-1 1st order and k = 2.3x10-4 s-1 2nd order and k = 4.3x10-4 s-1 2nd order and k = 2.3x10-4 s-1 zero and k = 4.3x10-4 s-1
Answers: 3

Do you know the correct answer?
N2O4 decomposes into NO2. At a certain temperature, the equilibrium pressures of NO2 and N2O4 are 1....
Questions in other subjects:

Geography, 25.04.2020 22:48

Geography, 25.04.2020 22:48



Mathematics, 25.04.2020 22:48

Social Studies, 25.04.2020 22:48




Computers and Technology, 25.04.2020 22:48