
Chemistry, 08.04.2020 20:07, Darkknighta26
The reaction 2 n2o5(g) → 4 no2(g) + o2(g) is found to obey the rate law: rate = k [n2o5]. does the reaction mechanism consist of only a single bimolecular step? 1. yes, it must 2. it might, but you cannot tell from the information given. 3. no, it cannot

Answers: 2
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 03:50, AysiaRamosLee
What is the temperature of one mole of helium gas at stp?
Answers: 3

Chemistry, 22.06.2019 14:20, montanolumpuy
7. in the cycle, a virus integrates its dna into the host's dna, and its dna is replicated when the host dna is replicated. a. infectious b. retroviral c. lysogenic d. lytic
Answers: 1

Chemistry, 22.06.2019 21:00, Janznznz4012
Once similarity and one difference between a mixture of elements and a mixture of compounds
Answers: 3
Do you know the correct answer?
The reaction 2 n2o5(g) → 4 no2(g) + o2(g) is found to obey the rate law: rate = k [n2o5]. does the r...
Questions in other subjects:



History, 29.06.2019 14:30

History, 29.06.2019 14:30


History, 29.06.2019 14:30


Mathematics, 29.06.2019 14:30


Mathematics, 29.06.2019 14:30


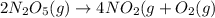
![Rate=k[N_2O_5]^1](/tpl/images/0589/8713/ae447.png)
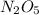 , it is a complex reaction.
, it is a complex reaction.



