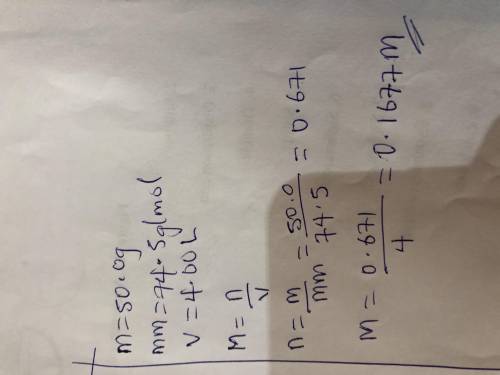50.0 grams of KCl is dissolved in water to make a 4.00 L
solution. What is the molarity of the...

Chemistry, 08.04.2020 19:03, SoccerdudeDylan
50.0 grams of KCl is dissolved in water to make a 4.00 L
solution. What is the molarity of the solution? (Molar mass of
KCl = 74.5 g/mol) ___ M ( Round your answer to the
appropriate number of four significant figures)

Answers: 1
Other questions on the subject: Chemistry

Chemistry, 21.06.2019 14:10, coralstoner6793
Which of these will change if the air in aclosed bottle is heated? abcdthe mass of the airthe composition of the airthe air pressure in the bottlethe number of air molecules in the bottle
Answers: 3


Chemistry, 22.06.2019 10:30, Brookwiggington8814
What woukd most likely be the transmittance at a 0.70 m solution of solute a? a) 7.6%b) 1.1%c)4.0%d)4.6%
Answers: 1

Chemistry, 22.06.2019 13:00, cnfndbxbfbdb2031
Which of the following are good traits of a hypothesis? it will be able to be testedit can predict an outcomeit will explain the observationsall of these
Answers: 2
Do you know the correct answer?
Questions in other subjects:


Advanced Placement (AP), 16.04.2021 22:00

Mathematics, 16.04.2021 22:00



Mathematics, 16.04.2021 22:00


Mathematics, 16.04.2021 22:00


Chemistry, 16.04.2021 22:00