Chemistry, 08.04.2020 05:01, markitakimbrough69
A chemist prepares a solution by adding 293 mgmg of K2Cr2O7K2Cr2O7 (MWMW = 294.19 g/molg/mol ) to a volumetric flask, and then adding water until the total volume of the contents of the flask reaches the calibration line that indicates 250 mLmL . Determine the molarity of the prepared solution.

Answers: 1
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 03:20, Richwave17
Which type of substance ionizes partially and gives off hydrogen ions when dissolved in water? a. strong acid b. strong base c. weak acid d. weak base
Answers: 1

Chemistry, 22.06.2019 04:00, eborkins
Seltzer water is created by placing water under pressure with carbon dioxide gas. which of the following statements best describe seltzer water: a. the solution will be slightly acidic b. the solution will be slightly basic. the solution will be strongly acidic. d. the solution will be strongly basic. e. the solution will be neutral
Answers: 3

Chemistry, 22.06.2019 04:10, tishfaco5000
Answer from each drop-down menu. e characteristics of a borane molecule (bh). the lewis structure and table of electronegativities are given olecular shape is and the molecule is reset next erved. search e a
Answers: 2

Chemistry, 22.06.2019 06:30, irvinbhangal2
What effect might melting sea ice have for people who live in coastal areas?
Answers: 1
Do you know the correct answer?
A chemist prepares a solution by adding 293 mgmg of K2Cr2O7K2Cr2O7 (MWMW = 294.19 g/molg/mol ) to a...
Questions in other subjects:

History, 03.06.2020 04:59

Mathematics, 03.06.2020 04:59


History, 03.06.2020 04:59







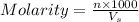
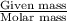
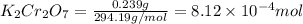
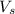 = volume of solution in ml = 250 ml
= volume of solution in ml = 250 ml