
C6H12O6 (s) + NH3 (g) -> C3H8O3 (l) + C2H6O(l) +CH1.83O0.56N0.17 (s) + CO2(g) + H2O Note that 1 mole glucose produces 0.5 mol glycerol and that the ratio of NH3 to H20 is 1:1. During preparation , you accidentally add too much glucose. However, you decide to let the bioreactor run anyway. After the reaction goes to completion at least one of the starting reactants is used up, you discover that 1.4 lb-m ethanol had been produced. How much heat had been produced and subsequently removed from the system. How much glucose did you initially add to the reactor?

Answers: 2
Other questions on the subject: Chemistry

Chemistry, 21.06.2019 22:00, carlybeavers50
The graph above shows how the price of cell phones varies with the demand quantity. the equilibrium price for cell phones is where both supply and demand quantities equal $100, 5,000 5,000, $100
Answers: 2

Chemistry, 22.06.2019 00:10, bossboybaker
Select the correct answer. which phrase correctly describes temperature? o a. average rotational kinetic energy of the particles in an object o b. average energy of the particles in an object c. average translational kinetic energy of the particles in an object od. all energy possessed by the particles in an object
Answers: 1

Chemistry, 22.06.2019 04:30, salvadorperez26
Suppose that during that icy hot lab 65,000 j of energy were transferred to 450 g of water at 20°c what would have have been the final temperature of the water
Answers: 2

Chemistry, 22.06.2019 08:30, masontdavis
Draw the skeletal structures of two different molecules that are each made of 5 carbon atoms and 12 hydrogen atoms.
Answers: 1
Do you know the correct answer?
C6H12O6 (s) + NH3 (g) -> C3H8O3 (l) + C2H6O(l) +CH1.83O0.56N0.17 (s) + CO2(g) + H2O Note that 1 m...
Questions in other subjects:



English, 20.09.2020 06:01


Engineering, 20.09.2020 06:01


Mathematics, 20.09.2020 06:01

Mathematics, 20.09.2020 06:01

Mathematics, 20.09.2020 06:01


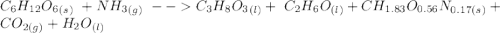
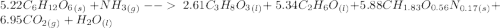
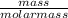
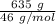
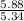 moles of S. cerevisiae formed.
moles of S. cerevisiae formed.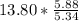 moles of S. cerevisiae formed
moles of S. cerevisiae formed



