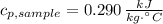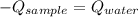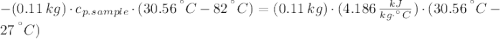Chemistry, 08.04.2020 02:09, lollipop83
A 110.0-g sample of metal at 82.00°C is added to 110.0 g of H O(l) at 27.00°C in an insulated container. The temperature rises to 30.56°C. Neglecting the heat capacity of the container, what is the specific heat of the metal? The specific heat of H O(l) is 4.18 J/(g ∙ °C).

Answers: 3
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 07:00, maryjane8872
Which set of characteristics best describes igneous rock? a) largest type of rock, made of organic matter, hardest type of rock b) least abundant type of rock, made of other rocks, made mostly of minerals c) found on all continents, contains wavy bands of stripes, contains fossils d) most abundant type in earth's crust, made of magma/lava, contains no fossils
Answers: 1

Chemistry, 22.06.2019 08:00, celestemaria0727
What are the similarities of physical and chemical change ?
Answers: 1
Do you know the correct answer?
A 110.0-g sample of metal at 82.00°C is added to 110.0 g of H O(l) at 27.00°C in an insulated contai...
Questions in other subjects:

Mathematics, 17.10.2020 04:01

Physics, 17.10.2020 04:01

Biology, 17.10.2020 04:01

Mathematics, 17.10.2020 04:01

Mathematics, 17.10.2020 04:01


Mathematics, 17.10.2020 04:01

English, 17.10.2020 04:01

Medicine, 17.10.2020 04:01

Mathematics, 17.10.2020 04:01