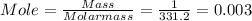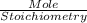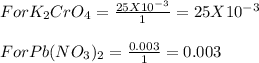4. When aqueous solutions of lead(II) ion are treated with potassium chromate solution, a bright yellow precipitate of lead(II) chromate, PbCrO4, forms. How many grams o lead chromate form when a 1.00-g sample of Pb(NO3)2 is added to 25.0mL of 1.00M K2CrO4 solution

Answers: 3
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 11:20, Jessicadiaz8602
Sodium nitrite (nano2) reacted with 2−iodooctane to give a mixture of two constitutionally isomeric compounds of molecular formula c8h17no2 in a combined yield of 88%. draw reasonable structures for these two isomers. click the "draw structure" button to launch the drawing utility. place the two compounds in the appropriate boxes below.
Answers: 1



Chemistry, 22.06.2019 20:20, carcon2019
Nitric acid can be formed in two steps from the atmospheric gases nitrogen and oxygen, plus hydrogen prepared by reforming natural gas. in the first step, nitrogen and hydrogen react to form ammonia: (g) (g) (g) in the second step, ammonia and oxygen react to form nitric acid and water: (g) (g) (g) (g) calculate the net change in enthalpy for the formation of one mole of nitric acid from nitrogen, hydrogen and oxygen from these reactions. round your answer to the nearest .
Answers: 3
Do you know the correct answer?
4. When aqueous solutions of lead(II) ion are treated with potassium chromate solution, a bright yel...
Questions in other subjects:


History, 24.04.2020 20:21

Mathematics, 24.04.2020 20:21





Biology, 24.04.2020 20:21

Mathematics, 24.04.2020 20:21