
Chemistry, 07.04.2020 23:59, ryliepeloquinf
A laser is emitting photons with a wavelength of 639.8 nm. What is the energy for 1 mole of these photons? For Planck's constant, use a value of 6.626x10-34 J s. Use units of kJ/mol. Report just the number, not the units.

Answers: 2
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 07:30, reaperqueen21
Compare and contrast the bohr model and the electron cloud models of the atom.
Answers: 1

Chemistry, 22.06.2019 09:00, angelrenee2000
What term is missing from the central region that describes hypotheses, theories, and laws? popular predictable mathematical falsifiable
Answers: 2

Do you know the correct answer?
A laser is emitting photons with a wavelength of 639.8 nm. What is the energy for 1 mole of these ph...
Questions in other subjects:

History, 16.10.2020 09:01

Mathematics, 16.10.2020 09:01


History, 16.10.2020 09:01


Mathematics, 16.10.2020 09:01


Computers and Technology, 16.10.2020 09:01



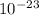
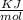
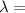 639.8 ×
639.8 × 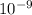 m
m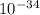 J sec
J sec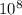 meter per second
meter per second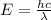
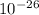
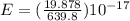
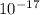 Joule per mole
Joule per mole



