
Chemistry, 07.04.2020 23:26, gevaughn600
If the caffeine concentration in a particular brand of soda is 2.43 mg/oz, drinking how many cans of soda would be lethal? Assume that 10.0 g of caffeine is a lethal dose, and there are 12 oz in a can.

Answers: 1
Other questions on the subject: Chemistry

Chemistry, 21.06.2019 20:30, Garciaapril1597
An exothermic reaction is conducted in an insulated calorimeter filled with water. the calorimeter is then sealed so that there is no heat exchanged between the contents of the container and the surrounding air. which of the following statements is true about the reaction?
Answers: 1

Chemistry, 22.06.2019 00:10, mpchop
According to the diagram; a) identify the anode of the cell and write the half-reaction that occurs there b) write the overall equation for the reaction that occurs as the cell operates c) calculate the value of the standard cell potential ,e cell. d)write the shorthand notation of the cell above e)indicate the flow of the electrons on the diagram
Answers: 3

Chemistry, 22.06.2019 04:00, heavyhearttim
4. absorption has the highest risk of overdose due to increased potency. a. rectal b. oral c. transdermal d. intranasal
Answers: 2

Chemistry, 22.06.2019 09:00, tbiles99
What type of energy do chemical bonds have? what type of energy is it converted to during chemical reactions? question 15 options: chemical bonds have kinetic energy, which is converted to potential energy during chemical reactions. chemical bonds have electric energy, which is converted to potential energy during chemical reactions. chemical bonds have heat energy, which is converted to kinetic energy during chemical reactions. chemical bonds have potential energy, which is converted to heat energy during chemical reactions.
Answers: 1
Do you know the correct answer?
If the caffeine concentration in a particular brand of soda is 2.43 mg/oz, drinking how many cans of...
Questions in other subjects:

Mathematics, 16.04.2021 22:30

Mathematics, 16.04.2021 22:30

Mathematics, 16.04.2021 22:30

Mathematics, 16.04.2021 22:30




History, 16.04.2021 22:30


Mathematics, 16.04.2021 22:30


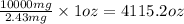
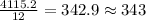 cans
cans



